 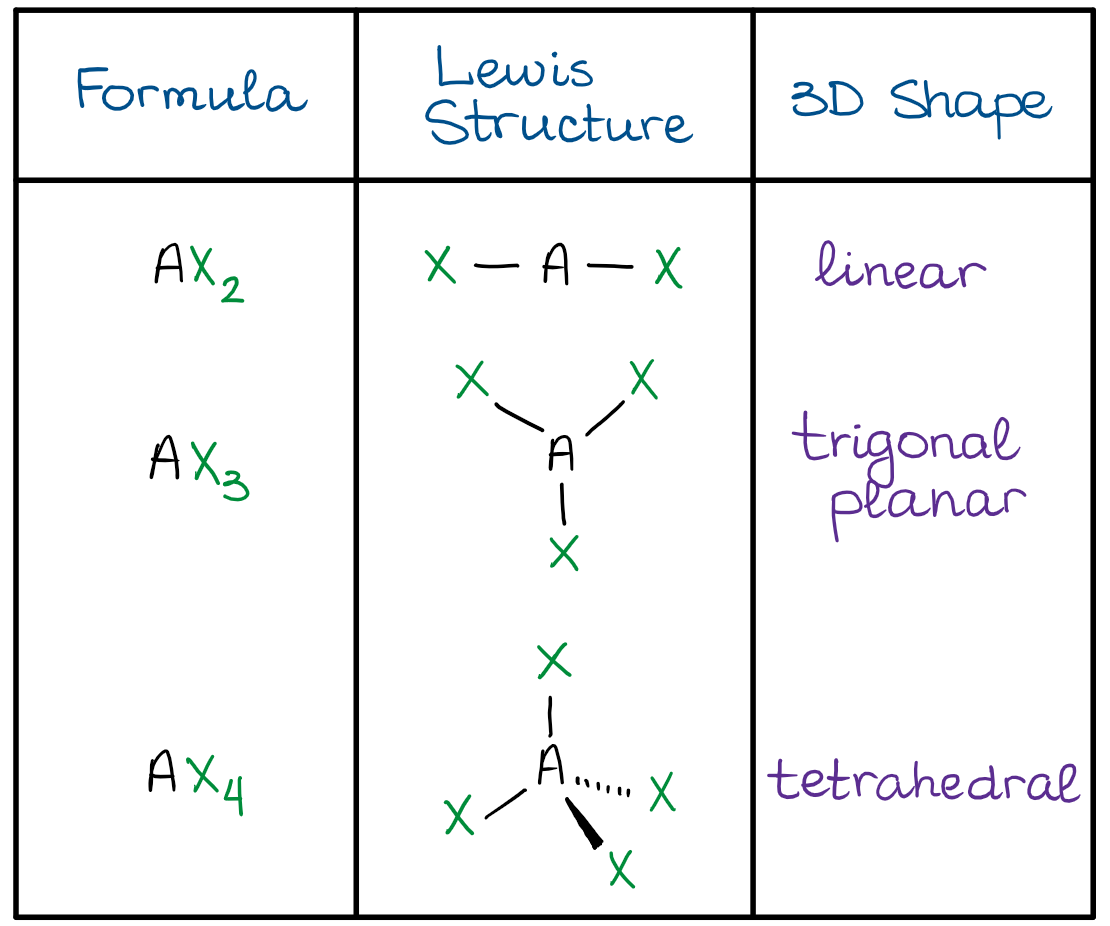
A half-full “s” orbital and a “d” subshell with 5 or 10 is more stable than following the Aufbau Principle. There are a few exceptions to the rules listed above when filling electron configurations. The noble gas method uses a noble gas (the far right column) to represent the inner, or core, electrons and just shows the outer level of electrons using the same method as spectroscopic. An up arrow and a down arrow have different “spins.” The spectroscopic method uses superscripts to show the number of electrons in a subshell (specific orbitals are not shown). The Boxes and Arrows method uses boxes to show orbitals and arrows to signify electrons. The Pauli Exclusion Principle states that when electrons do share an orbital, they must be of different “spin.” Hund’s Rule says that when placing electrons in orbitals of equal energy, place one in each orbital before doubling up in order to arrive at the lowest energy configuration. Determine the hybridization of the central atom (7). Predict the actual geometry of the molecule or ion (6). Predict the electronic geometry using all areas of electron density (or, effective electron pairs) and the ideal bond angles associated with this geometry (5). See the chart below for more information on how they are named depending on the number of lone pairs the molecule has. Determine the number of lone electron pairs on the central atom (4). Use the periodic table as a guide (read left to right): When the electron groups are all bond pairs, they are named exactly like the electron-group geometry. The Aufbau principle states that energy levels must be filled from the lowest to the highest and you may not move on to the next level unless the previous level is full. The approach was commonly referred to as VSEPR. Ronald Gillespie and Ronald Nyholm then developed the model into their theory published in 1957 they are considered the developers of the VSEPR theory. The model was proposed by Nevil Sidgwick and Herbert Powell in 1940. 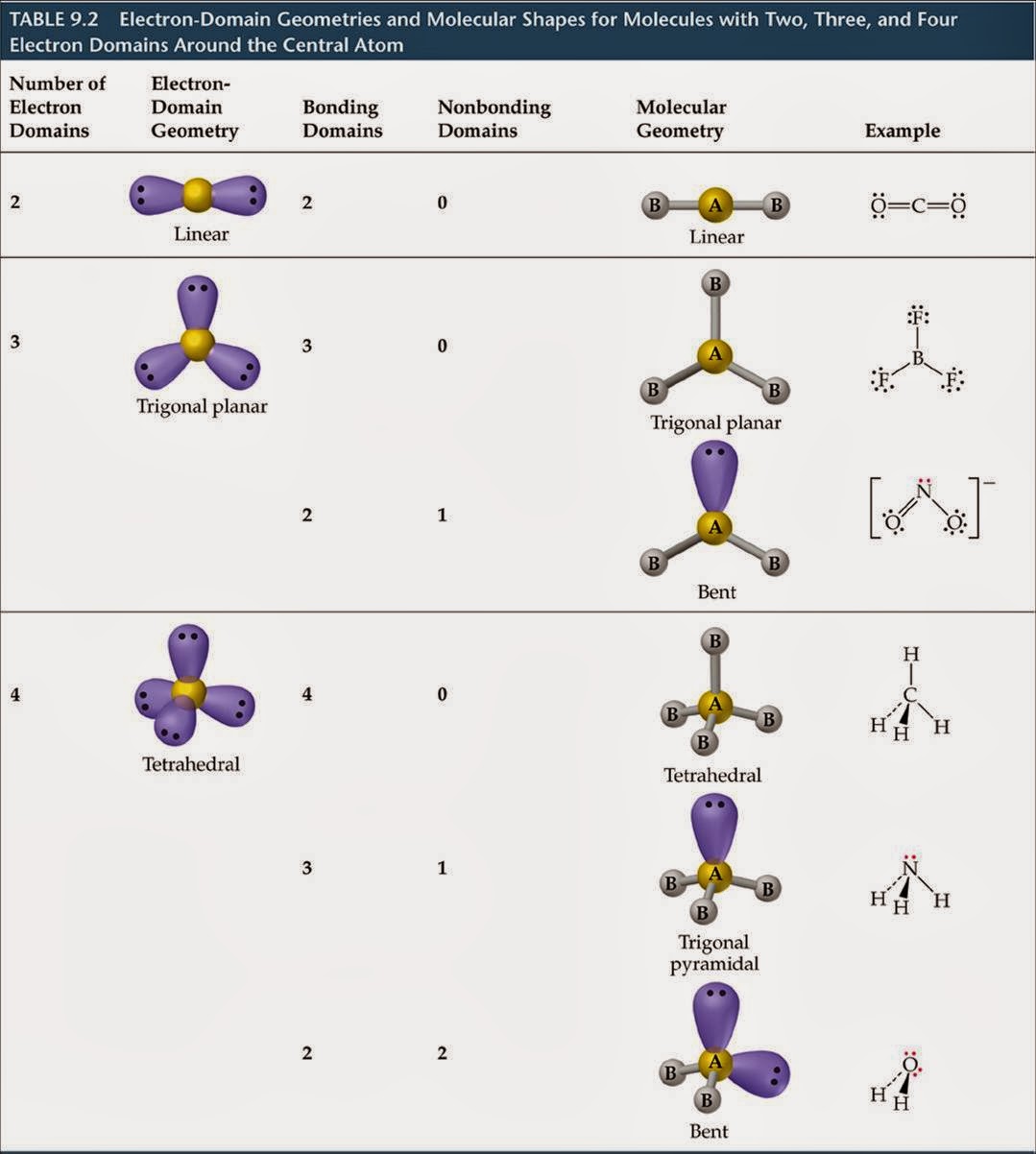
Rules for writing electron configurations VSEPR is an acronym that stands for valence shell electron pair repulsion. This video follows up on two videos introducing molecular and electron geometry. The subshells are further sub-divided into orbitals (s has 1 orbital, p has 3 orbitals, d has 5 orbitals and f has 7 orbitals). In this video we learn how to use electron and molecular geometry charts. The d is higher energy and begins in level 3 and the f is even higher energy and begins in level 4. Specifically, VSEPR models look at the bonding and molecular geometry of organic molecules and polyatomic ions. The p subshell is higher energy and therefore doesn’t begin until level 2. The valence shell electron pair repulsion model is often abbreviated as VSEPR (pronounced " vesper" ) and is a model to predict the geometry of molecules. The s subshell is the lowest energy and begins in level 1. These energy levels are sub-divided into subshells (labeled s, p, d or f). The number of electrons can be determined knowing the atomic number and charge.Įlectrons are placed in energy levels. The charge of the atom or ion is equal to the number of protons – the number of electrons. The atomic number is equal to the number of protons. Audiobooks for 40+ Courses in Science and Math (Lite Edition)Įlectrons are involved in bonding, and therefore, their arrangement in an atom is very important.Ītoms are composed of protons, neutrons and electrons.A table with four rows and six columns is shown. \): The basic electron-pair geometries predicted by VSEPR theory maximize the space around any region of electron density (bonds or lone pairs). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
